Lupin Launches Mesalamine Extended-Release Capsules - Authorized Generic to Apriso®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Mesalamine Extended-Release Capsules. Lupin's Mesalamine extended-release capsules are the authorized generic of Bausch Health Companies' Apriso® extended-release capsules. Mesalamine extended-release capsules are indicated for the maintenance of remission of ulcerative colitis in patients 18 years of age and older.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Mycophenolic Acid Delayed-Release Tablets USP - AB rated to Myfortic®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Mycophenolic Acid Delayed-Release Tablets USP. Lupin's Mycophenolic acid delayed-release tablets are the AB-rated generic equivalent of Novartis AG Corporation's Myfortic® delayed-release tablets. Mycophenolic acid tablets are an antimetabolite immunosuppressant indicated for prophylaxis of organ rejection in adult patients receiving kidney transplants and in pediatric patients at least 5 years of age and older who are at least 6 months post kidney transplant. Also indicated for use in combination with cyclosporine and corticosteroids.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Betamethasone Dipropionate Ointment USP (Augmented) 0.05% - AB rated to Diprolene®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Betamethasone Dipropionate Ointment USP (Augmented) 0.05%. Lupin's Betamethasone Dipropionate Ointment USP (Augmented) 0.05% is the AB-rated generic equivalent of Merck Sharp Dohme's Diprolene®. This product is a corticosteroid indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses in patients 13 years of age and older.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||||
Lupin Launches Naproxen and Esomeprazole Magnesium Delayed-Release Tablets - Authorized Generic to Vimovo®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Naproxen and Esomeprazole Magnesium Delayed-Release Tablets. Lupin's Naproxen and esomeprazole magnesium delayed-release tablets are the authorized generic of Horizon's Vimovo®. This product is a combination of naproxen, a non-steroidal anti-inflammatory drug (NSAID), and esomeprazole magnesium, a proton pump inhibitor (PPI) indicated in adult and adolescent patients 12 years of age and older weighing at least 38 kg, requiring naproxen for symptomatic relief of arthritis and esomeprazole magnesium to decrease the risk of developing naproxen-associated gastric ulcers. (reference Package Insert for further information).
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Ethacrynic Acid Tablets USP - AB rated to Edecrin®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Ethacrynic Acid Tablets USP. Lupin's Ethacrynic acid tablets are the AB-rated generic equivalent of Bausch Health Americas, Inc.'s Edecrin®. Ethacrynic acid tablets are indicated for the treatment of edema when an agent with greater diuretic potential than those commonly employed is required.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Moxifloxacin Ophthalmic Solution USP 0.5% - AT2 rated to Moxeza®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Moxifloxacin Ophthalmic Solution USP 0.5%. Lupin's Moxifloxacin ophthalmic solution is the AT2-rated generic equivalent of Alcon Pharmaceuticals Limited's Moxeza®. Moxifloxacin ophthalmic solution is a topical fluoroquinolone anti-infective indicated for the treatment of bacterial conjunctivitis caused by susceptible strains of certain organisms (reference Package Insert for further information).
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Oxycodone Hydrochloride Tablets USP - AB rated to Roxicodone®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Oxycodone Hydrochloride Tablets USP. Lupin's Oxycodone hydrochloride tablets are the AB-rated generic equivalent of Mallinckrodt Inc.'s Roxicodone® tablets. Oxycodone hydrochloride tablets are indicated for the management of pain severe enough to require an opioid analgesic and for which alternative treatments are inadequate.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Doxercalciferol for Injection - AP rated to Hectorol®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Doxercalciferol Injection. Lupin's Doxercalciferol Injection is the AP-rated generic equivalent of Sanofi Genzyme's Hectorol®. Doxercalciferol injection is indicated for the treatment of secondary hyperparathyroidism in adult patients with CKD on dialysis.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Hydrocortisone Valerate Cream USP 0.2% - AB rated to Westcort®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Hydrocortisone Valerate Cream USP 0.2%. Lupin's Hydrocortisone valerate cream is the AB-rated generic equivalent of Sun Pharmaceuticals Inc. Westcort® cream. Hydrocortisone valerate cream is a medium potency corticosteroid indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid responsive dermatoses in adult patients.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Potassium Chloride for Oral Solution USP 20 mEq - AA rated to Potassium Chloride for Oral Solution 20 mEq®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Potassium Chloride for Oral Solution USP 20 mEq. Lupin's Potassium Chloride for oral solution is the AA-rated generic equivalent of Pharma Research Software Solution, LLC's Potassium Chloride for Oral Solution, 20 mEq. Potassium chloride for oral solution 20 mEq is a potassium salt indicated for the treatment and prophylaxis of hypokalemia with or without metabolic alkalosis, in patients for whom dietary management with potassium-rich foods or diuretic dose reduction is insufficient.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Fluoxetine Tablets USP - AB rated to Prozac®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Fluoxetine Tablets USP. Lupin's Fluoxetine tablets are the AB-rated generic equivalent of Eli Lilly and Co.'s Prozac® tablets. Fluoxetine tablets are selective serotonin reuptake inhibitor indicated for acute and maintenance treatment of Major Depressive Disorder (MDD), Obsessive Compulsive Disorder (OCD), Bulimia Nervosa, and Panic Disorder, with or without agoraphobia.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Sildenafil Tablets USP - AB rated to Viagra®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Sildenafil Tablets USP. Lupin's Sildenafil tablets are the AB-rated generic equivalent of Pfizer Inc.'s Viagra® tablets. Sildenafil tablets are phosphodiesterase-5 (PDE5) inhibitors indicated for the treatment of erectile dysfunction (ED).
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||||
Lupin Launches Mycophenolate Mofetil Capsules USP 250mg - AB rated to CellCept®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Mycophenolate Mofetil Capsules USP 250mg. Lupin's Mycophenolate Mofetil capsules are the AB-rated generic equivalent of Hoffman-La Roche Inc. CellCept® capsules. Mycophenolate Mofetil capsules are an antimetabolite immunosuppressant indicated for the prophylaxis of organ rejection in recipients of allogeneic kidney, heart or liver transplants, and should be used in combination with other immunosuppressants.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Cefixime Capsules - AB rated to Suprax®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Cefixime capsules. Lupin's Cefixime capsules are the authorized generic equivalent of Lupin Limited's Suprax® capsules. Cefixime capsules is an antibacterial drug indicated in the treatment of adults and pediatric patients six months and older with the following infections: Uncomplicated Urinary Tract Infections, Otitis Media, Pharyngitis and Tonsillitis, Acute Exacerbations of Chronic Bronchitis, and Uncomplicated Gonorrhea (cervical/ urethral).
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for orderisng information.
|
|
|
|||||||
Lupin Launches Imatinib Mesylate Tablets - ABrated to Gleevec®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Imatinib Mesylate Tablets. Lupin's Imatinib Mesylate tabletsarethe AB-rated generic equivalent ofNovartis Pharmnaceuticals Gleevec® tablets. Imatinib Mesylate tablets are kinase inhibitors indicated for the treatment ofNewly Diagnosed Philadelphia Positive Chronic Myeloid Leukemia (Ph+ CML), Ph+ CML in Blast Crisis (BC), Accelerated Phase (AP) or Chronic Phase (CP) After Interferon-alpha (IFN) Therapy, Adult patients with Ph+ Acute Lymphoblastic Leukemia (ALL), Myelodysplastic/ Myeloproliferative Diseases (MDS/MPD), Aggressive Systemic Mastocytosis (ASM), Hypereosinophilic Syndrome (HES) and/or Chronic Eosinophilic Leukemia (CEL), and Dermatofibrosarcoma Protuberans (DFSP).
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Budesonide Inhalation Suspension 0.5 mg/2 mL - AN rated to Pulmicort Respules®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Budesonide Inhalation Suspension 0.5 mg/2 mL. Lupin's Budesonide inhalation suspension hydrochloride is the AN-rated generic equivalent of AstraZeneca's Pulmicort Respules®. Budesonide inhalation suspension is an inhaled corticosteroid indicated for maintenance treatment of asthma and as prophylactic therapy in children 12 months to 8 years of age.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Clobazam Tablets - AB rated to Onfi®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Clobazam tablets. Lupin's Clobazam tablets is the AB-rated generic equivalent of Lundback Pharms LLC's Onfi® tablets. Clobazam is benzodiazepine indicated for adjunctive treatment of seizures associated with Lennox-Gastaut syndrome (LGS) in patients 2 years of age or older.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Fluoxetine Tablets USP 60mg - AB rated to Fluoxetine Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Fluoxetine Tablets USP 60mg. Lupin's Fluoxetine tablets are the AB-rated generic equivalent of Alvogen Group Holdings 3 LLC's Fluoxetine tablets 60mg. Fluoxetine tablets are selective serotonin reuptake inhibitors (SSRI) indicated for the treatment of Major Depressive Disorder (MDD), Obsessive Compulsive Disorder (OCD), Bulimia Nervosa, and Panic Disorder, with or without agoraphobia.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for orderisng information.
|
|
|
|||||||||
Lupin Launches Testosterone Gel 1.62% - AB rated to AndroGel®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Testosterone Gel 1.62%. Lupin's Testosterone gel 1.62% is the AB-rated generic equivalent of AbbVie Inc.'s AndroGel® 1.62%. Testosterone gel 1.62% is indicated for replacement therapy in males for conditions associated with deficiency or absence of endogenous testosterone.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for orderisng information.
|
|
|
|||||||||
Lupin Launches Fluocinonide Ointment USP 0.05% - AB rated to Lidex®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Fluocinonide Ointment USP 0.05%. Lupin's Fluocinonide ointment USP 0.05% is the AB-rated generic equivalent of County Line Pharms Lidex® ointment 0.05%. Fluocinonide ointment USP 0.05% is indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Levothyroxine Sodium Tablets USP - AB2 rated to Synthroid®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Levothyroxine Sodium Tablets USP. Lupin's Levothyroxine sodium tablets is the AB2-rated generic equivalent of AbbVie Inc.'s Synthroid® tablets. Levothyroxine is indicated for Hypothyroidism - as replacement therapy in primary (thyroidal), secondary (pituitary), and tertiary (hypothalamic) congenital or acquired hypothyroidism and for Pituitary Thyrotropin Thyroid-Stimulating Hormone, TSH) Suppression - as an adjunct to surgery and radioiodine therapy in the management of thyrotropin-dependent well-differentiated thyroid cancer.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Ranolazine Extended-Release Tablets - AB rated to Ranexa®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Ranolazine Extended-Release Tablets. Lupin's Ranolazine Extended-Release tablets are the AB-rated generic equivalent of Gilead Sciences, Inc.'s Ranexa® extended-release tablets. Ranolazine extended-release tablets are indicated for the treatment of chronic angina and may be used with beta-blockers, nitrates, calcium channel blockers, antiplatelet therapy, lipid-lowering therapy, ACE inhibitors, and angiotensin receptor blockers.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Minocycline Hydrochloride Extended-Release Tablets USP, 55mg - AB rated to Solodyn®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Minocycline Hydrochloride Extended-Release Tablets USP, 55mg. Lupin's Minocycline hydrochloride extended-release tablets are the AB-rated generic equivalent of Bausch Health's Solodyn® extended-release tablets 55mg. Minocycline hydrochloride extended-release tablets are indicated to treat only inflammatory lesions of non-nodular moderate to severe acne vulgaris in patients 12 years of age and older.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Tadalafil Tablets USP 20mg - AB2 rated to Adcirca®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Tadalafil Tablets USP 20mg x 60. Lupin's Tadalafil tablets 20mg are the AB2-rated generic equivalent of Eli Lilly's Adcirca® tablets. Tadalafil tablets are phosphodiesterase 5 (PDE5) inhibitors indicated for the treatment of pulmonary arterial hypertension (PAH) to improve exerscise ability.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Clobazam Oral Suspension - AB rated to Onfi®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Clobazam oral suspension. Lupin's Clobazam oral suspension is the AB-rated generic equivalent of Lundback Pharms LLC's Onfi® oral suspension. Clobazam is benzodiazepine indicated for adjunctive treatment of seizures associated with Lennox-Gastaut syndrome (LGS) in patients 2 years of age or older.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Clomipramine Hydrochloride Capsules USP - AB rated to Anafranil®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Clomipramine Hydrochloride Capsules USP. Lupin's Clomipramine hydrochloride capsules USP is the AB-rated generic equivalent of SPECGX LLC's Anafranil® capsules. Clomipramine hydrochloride capsules USP is indicated for the treatment of obessions and compulsions in patients with Obsessive-Compulsive Disorder (OCD).
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Silodosin Capsules - AB rated to Rapaflo®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Silodosin capsules. Lupin's Silodosin capsules is the AB-rated generic equivalent of Allergan Salles LLC's Rapaflo® capsules. Silodosin is an alpha-1 adrenergic receptor antagonist, indicated for the treatment of the signs and symptoms of benign prostatic hyperplasia (BPH).
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Nystatin and Triamcinolone Acetonide Cream USP 100,000 units/gm; 0.1% - AT rated to Nystatin and Triamcinolone Acetonide Cream
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Nystatin and Triamcinolone Acetonide Cream USP 100,000 units/gm; 0.1%. Lupin's Nystatin and Triamcinolone Acetonide cream is the AT-rated generic equivalent of Taro Pharmaceuticals Nystatin and Triamcinolone Acetonide Cream. Nystatin and Triamcinolone Acetonide cream is a topical corticosteroid indicated for the treatment of cutaneous candidiasis.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Tetrabenazine Tablets - AB rated to Xenazine®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Tetrabenazine tablets. Lupin's Tetrabenazine tablets is the AB-rated generic equivalent of Valeant Pharmaceuticals Xenazine® Tablets. Tetrabenazine tablets is a vesicular monoamine transporter 2 (VMAT) indicated for the treatment of chorea associated with Huntington's disease.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Atovaquone Oral Suspension USP - AB rated to Mepron®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Atovaquone Oral Suspension USP. Lupin's Atovaquone oral suspension is the AB-rated generic equivalent of Glaxosmithkline LLC's Mepron® oral suspension. Atovaquone is a quinone antimicrobial drug indicated for the prevention of Pneumocystis jirovecii pneuomonia (PCP) in adults and adolescents aged 13 years and older who cannot tolerate trimethoprimsulfamethoxazole (TMP-SMX). It is also for the treatment of mild-to-moderate PCP in adults and adolescents aged 13 years and older who cannot tolerate TMP-SMX.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Clobetasol Propionate Ointment 0.05% - AB rated to Temovate®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Clobetasol Propionate Ointment 0.05%. Lupin's Clobetasol Propionate Ointment 0.05% is the AB-rated generic equivalent of Fougera Pharmaceuticals Inc.'s Temovate® Ointment. Clobetasol Propionate Ointment 0.05% is a super-high potency corticosteroid formulation indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid responsive dermatoses.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Potassium Chloride Oral Solution USP, 20 mEq/15mL (10%) and 40 mEq/15 mL (20%) - AA rated to Potassium Chloride Oral Solution
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Potassium Chloride Oral Solution USP, 20 mEq/15mL (10%) and 40 mEq/15 mL (20%). Lupin's Potassium Chloride Oral Solution is the AA-rated generic equivalent of Genus Life Sciences Inc.'s Potassium Chloride Oral Solution. Potassium Chloride Oral Solution is indicated for the treatment and prophylaxis of hypokalemia in patients for whom dietary management with potassium-rich foods or diuretic dose reduction are insufficient.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Hydrocortisone Butyrate Lotion 0.1% - AB rated to Locoid®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Hydrocortisone Butyrate Lotion 0.1%. Lupin's Hydrocortisone Butyrate Lotion 0.1% is the AB-rated generic equivalent of Leo Pharma's Locoid® Lotion 0.1%. Hydrocortisone Butyrate Lotion is an intermediate corticosteroid indicated for the topical treatment of mild to moderate atopic dermatitis in patient 3 months of age and older.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Clobetasol Propionate Cream USP 0.05% - AB1 rated to Temovate® Cream 0.05%
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Clobetasol Propionate Cream USP 0.05%. Lupin's Clobetasol Propionate Cream USP 0.05% is the AB1-rated generic equivalent of Fougera Pharmaceuticals Inc.'s Temovate® Cream 0.05%. Clobetasol Propionate Cream is a super-high potency corticosteroid indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
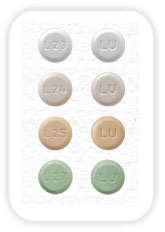
Lupin Launches Drospirenone, Ethinyl Estradiol and Levomefolate Calcium Tablets and Levomefolate Calcium Tablets, 3mg/0.02mg/0.451mg and 0.451mg - AB rated to Beyaz® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch Drospirenone, Ethinyl Estradiol and Levomefolate Calcium Tablets and Levomefolate Calcium Tablets, 3mg/0.02mg/0.451mg and 0.451mg. Lupin's Drospirenone, Ethinyl Estradiol and Levomefolate Calcium Tablets and Levomefolate Calcium Tablets are the AB-rated generic equivalent of Bayer Healthcare Pharmaceuticals Inc. (Bayer) Beyaz® Tablets and is an oral contraceptive indicated for use by females of reproductive potential to prevent pregnancy. Also indicated for the treatment of symptoms of premenstrual dysphoric disorder (PMDD).
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Fluocinonide Topical Solution USP 0.05% - AT rated to Lidex®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Fluocinonide Topical Solution USP 0.05%. Lupin's Fluocinonide Topical Solution USP 0.05% is the AT-rated generic equivalent of County Line Pharmaceuticals, LLC Lidex® Topical Solution 0.05%. Fluocinonide Topical Solution is a corticosteroid indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid responsive dermatoses.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Desoximetasone Topical Spray 0.25% - AT rated to Topicort®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Desoximetasone Topical Spray 0.25%. Lupin's Desoximetasone Topical Spray 0.25% is the AT-rated generic equivalent of Taro Pharmaceuticals U.S.A Inc.'s Topicort® Topical Spray 0.25%. Desoximetasone Topical Spray 0.25% is a corticosteroid indicated for the treatment of plaque psoriasis in patients 18 years of age or older.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Tobramycin Inhalation Solution USP - AN rated to Tobi®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Tobramycin Inhalation Solution USP. Lupin's Tobramycin Inhalation Solution is the AN-rated generic equivalent of Novartis Pharmaceuticals Corporation's Tobi®. Tobramycin Inhalation Solution is indicated for the management of cystic fibrosis patients with P. aeruginosa.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Methylergonovine Maleate Tablets USP - AB rated to Methergine®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Methylergonovine Maleate Tablets USP. Lupins Methylergonovine Maleate Tablets USP are the AB-rated generic equivalent of Novartis Pharmaceuticals Corporations Methergine® Tablets. Methylergonovine Maleate tablets are semi-synthetic ergot alkaloids used for the prevention and control of postpartum hemorrhage.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Clobetasol Propionate Spray 0.05% - AT rated to Clobex®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Clobetasol Propionate Spray 0.05%. Lupin's Clobetasol Propionate Spray 0.05% is the AT-rated generic equivalent of Galderma Laboratories, L.P.'s Clobex® Propionate Spray. Clobetasol propionate spray 0.05% is a super-high potent topical corticosteroid formulation indicated for the treatment of moderate to severe plaque psoriasis affecting up to 20% body surface area (BSA) in patients 18 years of age or older.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Carbidopa Tablets - AB rated to Lodosyn®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Carbidopa Tablets. Lupin's Carbidopa Tablets are the AB-rated generic equivalent of Aton Pharma Inc.'s Lodosyn® Tablets. Carbidopa tablets are dopa-decarboxylase inhibitors indicated for use with carbidopa-levodopa or with levodopa in the treatment of the symptoms of idiopathic Parkinson's disease (paralysis agitans), postencephalitic parkinsonism, and symptomatic parkinsonism, which may follow injury to the nervous system by carbon monoxide intoxication and/or manganese intoxication.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Tydemy® Tablets (Drospirenone, ethinyl estradiol and levomefolate calcium tablets, 3mg/0.03mg/0.451mg and levomefolate calcium tablets, 0.451mg) - AB rated to Safyral® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch Tydemy® Tablets (Drospirenone, ethinyl estradiol and levomefolate calcium tablets, 3mg/0.03mg/0.451mg and levomefolate calcium tablets, 0.451mg). Lupin's Tydemy® Tablets are the AB-rated generic equivalent of Bayer Healthcare Pharmaceuticals Inc. (Bayer) Safyral® Tablets and is an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Testosterone Topical Solution 30mg/1.5mL - AT rated to Axiron®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Testosterone Topical Solution 30mg/1.5mL. Lupin's Testosterone Topical Solution is the AT-rated generic equivalent of Eli Lilly and Company's Axiron®. Testosterone Topical Solution is indicated for replacement therapy in males for conditions associated with a deficiency or absence of endogenous testosterone.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Oseltamivir Phosphate for Oral Suspension - AB rated to Tamiflu® for Oral Suspension
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Oseltamivir Phosphate for Oral Suspension. Lupin's Oseltamivir Phosphate for Oral Suspension is the AB-rated generic equivalent of Hoffman La Roche Inc. Tamiflu® for Oral Suspension. Oseltamivir Phosphate for Oral Suspension is indicated for the treatment of acute, uncomplicated illness due to influenza A and B infection in patients 2 weeks of age and older who have been symptomatic for no more than 48 hours.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Memantine Hydrochloride Extended-Release Capsules - AB rated to Namenda XR® Capsules
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Memantine Hydrochloride Extended-Release Capsules. Lupin's Memantine Hydrochloride Extended-Release Capsules are the AB-rated generic equivalent of Forest Laboratories Inc. Namenda XR® Capsules. Memantine Hydrochloride Extended-Release Capsules are central nervous system agents indicated for the treatment of moderate to severe dementia of the Alzheimer's type.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Oseltamivir Phosphate Capsules - AB rated to Tamiflu® Capsules
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Oseltamivir Phosphate Capsules. Lupin's Oseltamivir Phosphate Capsules are the AB-rated generic equivalent of Hoffman La Roche Inc. Tamiflu® Capsules. Oseltamivir Phosphate Capsules are neuraminidase inhibitors indicated for the treatment of acute, uncomplicated illness due to influenza A and B infection in patients 2 weeks of age and older who have been symptomatic for no more than 48 hours.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Clonidine Hydrochloride Extended-Release Tablets - AB1 rated to Kapvay®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Clonidine Hydrochloride Extended-Release Tablets. Lupin's Clonidine Hydrochloride Extended-Release Tablets are the AB1-rated generic equivalent of Concordia Pharmaceuticals Inc.'s Kapvay® Extended-Release Tablets. Clonidine hydrochloride extended-release tablets are a centrally acting alpha2-adrenergic agonist indicated for the treatment of attention deficit hyperactivity disorder (ADHD) as monotherapy or as adjunctive therapy to stimulant medications.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
Lupin Launches Clobetasol Propionate Lotion 0.05% - AB rated to Clobex®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Clobetasol Propionate Lotion 0.05%. Lupin's Clobetasol Propionate Lotion 0.05% is the AB-rated generic equivalent of Galderma Laboratories, L.P.'s Clobex® Propionate Lotion. Clobetasol propionate lotion 0.05% is a corticosteroid indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses, in patients 18 years of age or older.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||||
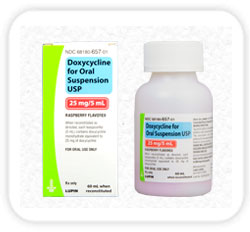
Lupin Launches Doxycycline Hyclate Tablets USP - AB rated to Vibra-Tabs®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Doxycycline Hyclate Tablets USP. Lupin's Doxycycline Hyclate Tablets USP is the AB-rated generic equivalent of Pfizer Labs Vibra-Tabs®. Doxycycline Hyclate Tablets are an antibiotic indicated to treat many different bacterial infections, such as acne, urinary tract infections, intestinal infections, eye infections, gonorrhea, chlamydia, periodontitis (gum disease), and others.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Flucytosine Capsules USP - AB rated to Ancobon®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Flucytosine Capsules USP. Lupin's Flucytosine Capsules USP are the AB-rated generic equivalent of Valeant Pharmaceuticals International's Ancobon® Capsules. Flucytosine Capsules are an antifungal agent indicated only in the treatment of serious infections caused by susceptible strains of Candida and/or Cryptococcus.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Doxycycline Hyclate Tablets USP - AB rated to Acticlate®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Doxycycline Hyclate Tablets USP. Lupin's Doxycycline Hyclate Tablets USP are the AB-rated generic equivalent of Aqua Pharms, LLC Acticlate® Tablets. Doxycycline Hyclate Tablets are tetracycline antibiotics indicated for the treatment of various sexually transmitted infections.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Hydrocodone Bitartrate and Acetaminophen Tablets USP - AA rated to Norco®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Hydrocodone Bitartrate and Acetaminophen Tablets USP. Lupin's Hydrocodone Bitartrate and Acetaminophen Tablets USP are the AA-rated generic equivalent of Allergan Pharmaceuticals' Norco® Tablets. Hydrocodone Bitartrate and Acetaminophen Tablets are an opioid / acetaminophen combination indicated for the relief of moderate to moderately severe pain.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Lanthanum Carbonate Chewable Tablets - AB rated to Fosrenol®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Lanthanum Carbonate Chewable Tablets. Lupin's Lanthanum Carbonate Chewable Tablets are the AB-rated generic equivalent of Shire LLC's Fosrenol® Chewable Tablets. Lanthanum Carbonate Chewable Tablets are phosphate binder indicated to reduce serum phosphate in patients with end stage renal disease (ESRD).
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Quetiapine Fumarate Extended-Release Tablets - AT rated to Seroquel XR®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Quetiapine Fumarate Extended-Release Tablets. Lupin's Quetiapine Fumarate Extended-Release Tablets are the AB-rated generic equivalent of AstraZeneca Pharmaceuticals LP (AstraZeneca) Seroquel XR® Tablets. Quetiapine Fumarate Extended-Release Tablets are an atypical antipsychotic indicated for the treatment of Schizophrenia, Bipolar I disorder, manic or mixed episodes, Bipolar disorder, depressive episodes and Major depressive disorder, adjunctive therapy with antidepressants.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Moxifloxacin Ophthalmic Solution USP, 0.5% - AT rated to Vigamox®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Moxifloxacin Ophthalmic Solution USP, 0.5%. Lupin's Moxifloxacin Ophthalmic Solution is the AT-rated generic equivalent of Alcon Pharmaceuticals Ltd.'s (Novartis) Vigamox®. Moxifloxacin Ophthalmic Solution is a topical fluoroquinolone anti-infective indicated for the treatment of bacterial conjunctivitis caused by susceptible strains of different organisms.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Bupropion Hydrochloride Extended-Release Tablets - AB rated to Wellbutrin XL® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Bupropion Hydrochloride Extended-Release Tablets. Lupin's Bupropion Hydrochloride Extended-Release Tablets are the AB-rated generic equivalent of GlaxoSmithKline, LLC.'s Wellbutrin XL® Tablets. Bupropion Hydrochloride Extended-Release Tablets are antidepressants indicated for major depressive disorder and for the prevention of seasonal affective disorder.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Desoximetasone Cream USP, 0.05% (AB rated to Topicort® LP Emollient Cream) and 0.25% (AB rated to Topicort® Cream)
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Desoximetasone Cream 0.25% (AB rated to Topicort® Cream) and 0.05% (AB rated to Topicort® LP Emollient Cream of Taro Pharmaceuticals North America). Desoximetasone Cream USP is indicated to treat inflammatory and itching caused by a number of skin conditions such as allergic reactions, eczema, and psoriasis.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Desoximetasone Ointment USP, 0.25% - AB rated to Topicort® Ointment
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Desoximetasone Ointment USP, 0.25%. Lupin's Desoximetasone Ointment USP, 0.25% is the AB-rated generic equivalent of Taro Pharmaceuticals, Inc.'s Topicort® Ointment. Desoximetasone Ointment USP, 0.25% is indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Desoximetasone Ointment USP, 0.05% - AB rated to Topicort® Ointment
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Desoximetasone Ointment USP, 0.05% x 60g. Lupin's Desoximetasone Ointment USP, 0.05% is the AB-rated generic equivalent of Taro Pharms North's Topicort® Ointment. Desoximetasone Ointment USP, 0.05% is indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Fayosim® Tablets (Levonorgestrel and Ethinyl Estradiol Tablets USP, 0.15 mg/0.02 mg; 0.15 mg/0.025 mg; 0.15 mg/0.03 mg and Ethinyl Estradiol Tablets USP, 0.01mg) - AB rated to Quartette© Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch Fayosim® Tablets (Levonorgestrel and Ethinyl Estradiol Tablets USP, 0.15 mg/0.02 mg; 0.15 mg/0.025 mg; 0.15 mg/0.03 mg and Ethinyl Estradiol Tablets USP, 0.01mg). Lupin's Fayosim® Tablets are the AB-rated generic equivalent of Teva Branded Pharmaceuticals Products R&D, Inc. (Teva) Quartette® Tablets and is an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Abacavir and Lamivudine Tablets USP - AB rated to Epzicom® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Abacavir and Lamivudine Tablets. Lupin's Abacavir and Lamivudine Tablets are the AB-rated generic equivalent of ViiV Healthcare Company's Epzicom® Tablets. Abacavir and Lamivudine Tablets are nucleoside analogue HIV-1 reverse transcriptase inhibitors indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Mibelas™ 24 Fe Tablets (Norethindrone Acetate and Ethinyl Estradiol Chewable Tablets, 1 mg/0.02 mg and Ferrous Fumarate Tablets, 75 mg) - AB rated to Minastrin® 24 Fe Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch Mibelas™ 24 Fe Tablets (Norethindrone Acetate and Ethinyl Estradiol Chewable Tablets, 1 mg/0.02 mg and Ferrous Fumarate Tablets, 75 mg). Lupin's Mibelas™ 24 Fe Tablets are the AB-rated generic equivalent of Allergan Pharmaceuticals International Limited (Allergan) Minastrin® 24 Fe Tablets and is an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Hydrocodone Bitartrate and Acetaminophen Tablets USP- AA rated to Xodol® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Hydrocodone Bitartrate and Acetaminophen Tablets USP. Lupin's Hydrocodone Bitartrate and Acetaminophen Tablets USP are the AA-rated generic equivalent of Victory Pharma, Inc.'s Xodol@reg; Tablets. Hydrocodone Bitartrate and Acetaminophen Tablets are an opioid / acetaminophen combination indicated for the relief of moderate to moderately severe pain.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Paroxetine HCl Extended-Release Tablets - AB rated to Paxil CR® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Paroxetine HCl Extended-Release Tablets. Lupin's Paroxetine HCl Extended-Release Tablets are the AB-rated generic equivalent of GlaxoSmithKline, LLC's Paxil CR® Tablets. Paroxetine HCl Extended-Release Tablets are antidepressants for the treatment of major depressive disorder.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Desvenlafaxine Succinate Extended-Release Tablets ' AB rated to Pristiq® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Desvenlafaxine Succinate Extended-Release Tablets. Lupin's Desvenlafaxine Succinate Extended-Release Tablets are the AB-rated generic equivalent of Wyeth Pharmaceuticals Pristiq® Tablets. Desvenlafaxine Succinate Extended-Release Tablets are serotonin and norepinephrine reuptake inhibitor (SNRI) indicated for the treatment of major depressive disorder (MDD).
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Norgestimate and Ethinyl Estradiol Tablets USP - AB rated to Ortho-Cyclen® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch Norgestimate and Ethinyl Estradiol Tablets USP, 0.25 mg/0.035 mg. Lupin's Norgestimate and Ethinyl Estradiol Tablets are the AB-rated generic equivalent of Janssen Pharmaceuticals® Ortho-Cyclen® Tablets and is an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Armodafinil Tablets - AB rated to Nuvigil® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Armodafinil Tablets. Lupin's Armodafinil Tablets are the AB-rated generic equivalent of Cephalon, Inc.'s Nuvigil® Tablets. Armodafinil Tablets are CNS stimulants indicated to improve wakefulness in adult patients with excessive sleepiness associated with obstructive sleep apnea (OSA), narcolepsy, or shift work disorder (SWD).
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Armodafinil Tablets. Lupin's Armodafinil Tablets are the AB-rated generic equivalent of Cephalon, Inc.'s Nuvigil® Tablets. Armodafinil Tablets are CNS stimulants indicated to improve wakefulness in adult patients with excessive sleepiness associated with obstructive sleep apnea (OSA), narcolepsy, or shift work disorder (SWD).
|
|
|
Lupin Launches Amabelz™ Tablets (Estradiol and Norethindrone Acetate Tablets USP), 0.5 mg/0.1 mg and 1 mg/0.5 mg' AB rated to Activella® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Amabelz® Tablets (Estradiol and Norethindrone Acetate Tablets USP), 0.5 mg/0.1 mg and 1 mg/0.5 mg. Lupin's Amabelz® Tablets USP are the AB-rated generic equivalent of Amneal Pharmaceuticals' Activella® Tablets and is a hormone replacement therapy indicated to treat symptoms of menopause and prevention of postmenopausal osteoporosis.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Norgestimate and Ethinyl Estradiol Tablets USP, 0.18mg/0.035mg, 0.215mg/0.035mg and 0.25mg/0.035mg - AB rated to Ortho Tri-Cyclen® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Norgestimate and Ethinyl Estradiol Tablets USP, 0.18mg/0.035mg, 0.215mg/0.035mg and 0.25mg/0.035mg. Lupin's Norgestimate and Ethinyl Estradiol Tablets USP are the AB-rated generic equivalent of Janssen Pharmaceuticals' Ortho Tri-Cyclen® Tablets and is an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Diclofenac Sodium Topical Solution 1.5% - AT rated to Pennsaid® 1.5% Topical Solution
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Diclofenac Sodium Topical Solution 1.5%. Lupin's Diclofenac Sodium Topical Solution is the AT-rated generic equivalent of Mallinckrodt's Pennsaid® 1.5% Topical Solution. Diclofenac Sodium Topical Solution is a nonsteroidal anti-inflammatory drug (NSAID) indicated for the treatment of signs and symptoms of osteoarthritis of the knee(s).
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Blisovi™ Fe 1.5/30 Tablets - AB rated to Loestrin® Fe 1.5/30 Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Blisovi™ Fe 1.5/30 Tablets (Norethindrone Acetate and Ethinyl Estradiol Tablets USP and Ferrous Fumarate Tablets 1.5 mg/0.03 mg). Lupin's Blisovi™ Fe1.5/30 Tablets are the AB-rated generic equivalent of Warner Chilcott's Loestrin® Fe 1.5/30 Tablets and is an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Blisovi™ Fe 1/20 Tablets - AB rated to Loestrin® Fe 1/20 Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Blisovi™ Fe 1/20 Tablets (Norethindrone Acetate and Ethinyl Estradiol Tablets USP and Ferrous Fumarate Tablets 1mg/0.02mg). Lupin's Blisovi™ Fe 1/20 Tablets are the AB-rated generic equivalent of Warner Chilcott's Loestrin® Fe 1/20 Tablets and is an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Levonorgestrel and Ethinyl Estradiol Tablets USP 0.05 mg/0.03 mg; 0.075 mg/0.04 mg and 0.125 mg/0.03 mg - AB rated to Trivora® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Levonorgestrel and Ethinyl Estradiol Tablets USP 0.05 mg/0.03 mg; 0.075 mg/0.04 mg and 0.125 mg/0.03 mg. Lupin's Levonorgestrel and Ethinyl Estradiol Tablets USP are the AB-rated generic equivalent of Actavis' Trivora® Tablets and is an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Donepezil Hydrochloride Tablets - AB rated to Aricpet® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Donepezil Hydrochloride Tablets. Lupin's Donepezil Hydrochloride Tablets are the AB-rated generic equivalent of Eisai R&D Management's Aricept® Tablets. Donepezil Hydrochloride Tablets are acetylcholinesterase inhibitors indicated for the treatment of mild to moderate dementia of the Alzheimer's type.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Fyavolv™ Tablets - AB rated to FemHRT® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Fyavolv™ (Norethindrone Acetate and Ethinyl Estradiol Tablets USP 0.5 mg/0.0025 mg; 1 mg/0.005 mg). Lupin's Fyavolv™ is the AB-rated generic equivalent of Warner Chilcott's FemHRT® and is a hormone replacement therapy indicated to treat symptoms of menopause and prevent osteoporosis.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Potassium Chloride ER Capsules USP ' AB rated to Micro-K® Capsules
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Potassium Chloride Extended-Release Capsules. Lupin's Potassium Chloride ER Capsules are the AB-rated generic equivalent of Nesher Pharmaceuticals, LLC's (a Zydus Pharmaceuticals Company) Micro-K® Capsules. Potassium Chloride ER Capsules are electrolyte replenishers indicated for treatment of patients with hypokalemia with or without metabolic alkalosis.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Metformin Hydrochloride Extended-Release Tablets USP - AB3 rated to Glumetza® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Metformin Hydrochloride Extended-Release Tablets. Lupin's Metformin Hydrochloride Extended-Release Tablets are the AB3-rated generic equivalent of Santarus, Inc.'s Glumetza® Tablets. Metformin Hydrochloride Extended-Release Tablets are antihyperglycemic drugs used to treat patients with high blood sugar levels that are caused by type 2 diabetes.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
|||||||
Lupin Launches Tri-Lo-Marzia™ Tablets - AB rated to Ortho Tri-Cyclen® Lo Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Tri-Lo-Marzia™ Tablets 0.180 mg/0.025 mg; 0.215 mg/0.025 mg and 0.250 mg/0.025 mg. Lupin's Tri-Lo- Marzia™ Tablets are the AB-rated generic equivalent of Janssen Pharmaceuticals' Ortho Tri- Cyclen® Lo Tablets and is an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
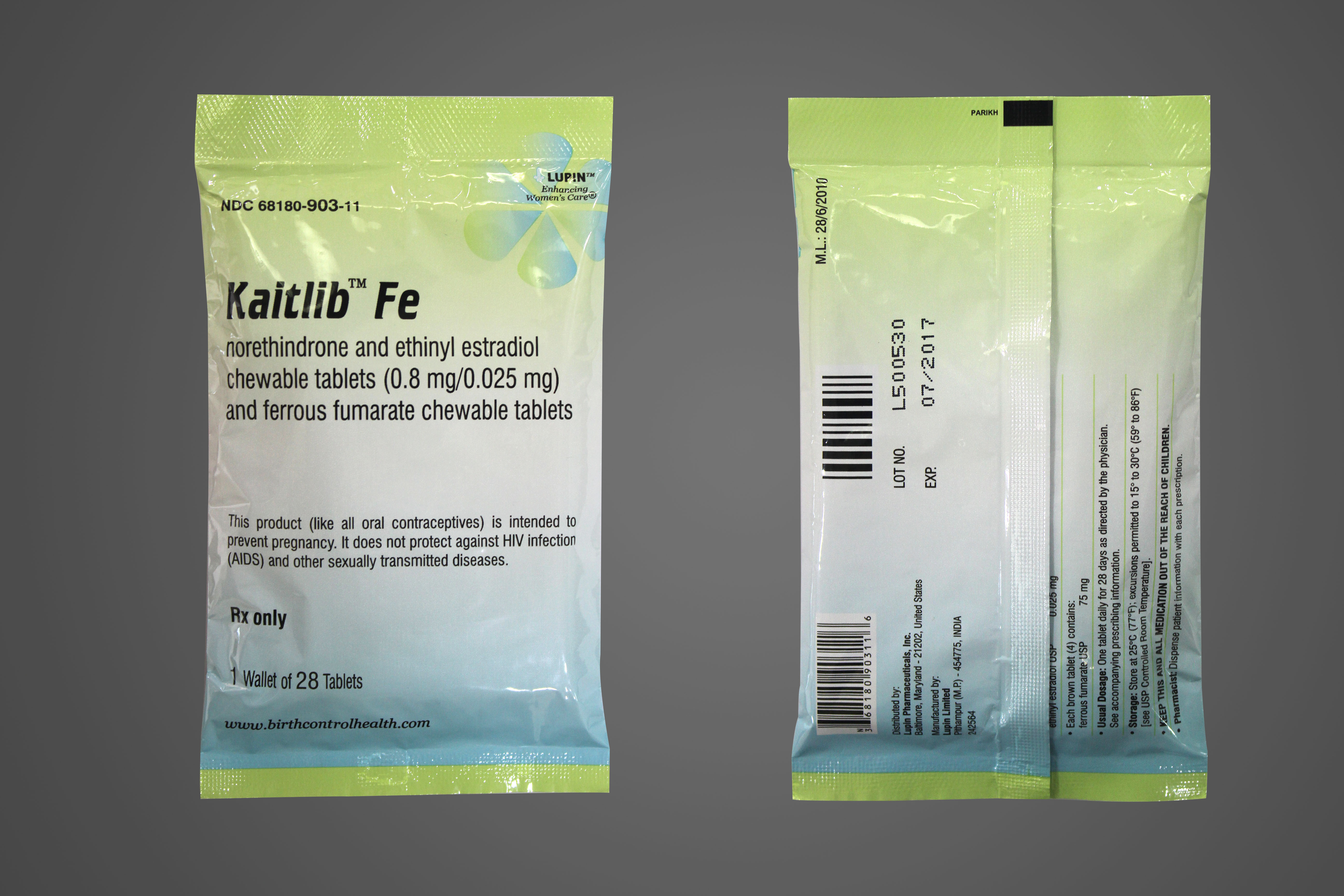
Lupin Launches Kaitlib™ Fe Tablets - AB rated to Generess® Fe Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Kaitlib™ Tablets (Norethindrone and Ethinyl Estradiol Chewable Tablets 0.8mg/0.025mg and Ferrous Fumarate Chewable Tablets). Lupin's Kaitlib™ Fe Tablets are the AB-rated generic equivalent of Actavis® Generess® Fe Tablets and are an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Blisovi™ 24 Fe Tablets- AB rated to Loestrin® 24 Fe Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Blisovi™24 Fe Tablets (Norethindrone Acetate and Ethinyl Estradiol USP (1mg/0.02mg), Ethinyl Estradiol Tablets USP (0.01mg) and Ferrous Fumarate Tablets). Lupin'sBlisovi™24 Fe Tablets arethe AB-rated generic equivalent of Warner Chilcott's Loestrin® 24Fe Tablets and is an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
Lupinhas already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Repaglinide and Metformin Hydrochloride Tablets - AB rated to PrandiMet® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Repaglinide and Metformin Hydrochloride Tablets. Lupin's Repaglinide and Metformin Hydrochloride Tablets are the AB-rated generic equivalent of Novo Nordisc's PrandiMet® Tablets. Repaglinide and Metformin Hydrochloride Tablets are anti-diabetics indicated for treatment of patients with high blood sugar levels caused by type 2 diabetes.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Bekyree™ Tablets - AB rated to Mircette® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch BeKyree™ Tablets (Desogestrel and Ethinyl Estradiol Tablets USP 0.15 mg/0.02 mg and Ethinyl Estradiol Tablets USP 0.01 mg). Lupin's BeKyree™ Tablets are the AB-rated generic equivalent of Teva's Mircette® Tablets and is an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Fenofibrate Tablets - AB rated to LoFibra® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Fenofibrate Tablets. Lupin's Fenofibrate Tablets are the AB-rated generic equivalent of Teva's Lofibra® Tablets. Fenofibrate Tablets are antihyperlipidemic agents indicated for treatment of patients with high cholesterol and high triglyceride levels.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Calcium Acetate Capsules - AB rated to PhosLo® Gel Capsules
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Calcium Acetate Capsules. Lupin's Calcium Acetate Capsules are the AB-rated generic equivalent of Fresenius's PhosLo® Gel Capsules. Calcium Acetate Capsules are a phosphate binder indicated for the control of Hyperphosphatemia (electrolyte disturbance in which there is an abnormally elevated level of phosphate in the blood) in patients with end stage renal disease (ESRD).
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Azithromycin Tablets USP - AB rated to Zithromax®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Azithromycin Tablets. Lupin's Azithromycin Tablets are the AB-rated generic equivalent of Pfizer's Zithromax® Tablets. Azithromycin Tablets USP is a macrolide antibiotic indicated for treatment of bacterial infections.Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
Lupin has already commenced shipment of the product in the U.S.
Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Quinine Sulfate Capsules USP- AB rated to Qualaquin®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Quinine Sulfate Capsules. Lupin's Quinine Sulfate Capsules are the AB-rated generic equivalent of Mutual's Qualaquin® Capsules. Quinine Sulfate Capsules USP are a cinchona alkaloid indicated for treatment of uncomplicated Plasmodium falciparum malaria.
Lupin has already commenced shipment of the product in the U.S.
Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Memantine HCl Tablets USP - AB rated to Namenda®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Memantine Hydrochloride (HCl) Tablets.
Lupin's Memantine HCl Tablets are the AB-rated generic equivalent of Forest Lab's Namenda® Tablets and is an N-methyl-D-aspartate (NMDA) receptor antagonist indicated for the treatment of moderate to severe dementia of the Alzheimer's type.
Lupin has already commenced shipment of the product in the U.S.
Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
||||||
Lupin Launches Amlodipine, Valsartan and Hydrochlorothiazide Tablets - AB rated to Exforge HCT®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Amlodipine, Valsartan and Hydrochlorothiazide Tablets.
Lupin's Amlodipine, Valsartan and Hydrochlorothiazide Tablets are the AB-rated generic equivalent of Novartis Exforge HCT® Tablets.
Amlodipine, Valsartan and Hydrochlorothiazide Tablets are indicated for the treatment of hypertension to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes, and myocardial infarctions. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions.
Lupin has already commenced shipment of the product in the U.S.
Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
||||||||
Lupin Exclusively Launches Bimatoprost Ophthalmic Solution 0.03%
Lupin Pharmaceuticals, Inc. is proud to announce the launch of Bimatoprost Ophthalmic Solution 0.03%. Lupin's Bimatoprost 0.03% is the first generic alternative to Allergan's Lumigan® to receive final FDA approval.
Bimatoprost Ophthalmic Solution 0.03% is a prostaglandin analog indicated for the reduction of elevated intraocular pressure in patients with open angle glaucoma or ocular hypertension.
Lupin has already commenced shipment of the product in the U.S.
Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Vancomycin Hydrochloride Capsules USP - AB rated to Vancocin®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Vancomycin Hydrochloride Capsules USP. Lupin's Vancomycin Hydrochloride Capsules USP are the AB-rated generic equivalent of Eli Lilly's Vancocin® Capsules.
Vancomycin Hydrochloride Capsules USP are indicated for the treatment of C. difficile associated diarrhea. Vancomycin Hydrochloride Capsules USP are also used for the treatment of enterocolitis caused by Staphylococcus aureus (including methicillin-resistant strains).
Lupin has already commenced shipment of the product in the U.S.
Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Amlodipine & Valsartan Tablets - AB rated to Exforge®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Amlodipine and Valsartan Tablets.
Lupin's Amlodipine and Valsartan Tablets are the AB-rated generic equivalent of Novartis Exforge® Tablets. Amlodipine and Valsartan Tablets is the combination tablet of Amlodipine, a dihydropyridine calcium channel blocker (DHP CCB), and Valsartan, an angiotensin II receptor blocker (ARB). Amlodipine and Valsartan Tablets are indicated for the treatment of hypertension, to lower blood pressure:
- In patients not adequately controlled on monotherapy.
- As initial therapy in patients likely to need multiple drugs to achieve their blood pressure goals.
Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Lamivudine Tablets - AB Rated to Epivir®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Lamivudine tablets,
150mg & 300mg. Lupin's Lamivudine tablets are the AB-rated generic equivalent of Viiv
Healthcare's Epivir®. Lamivudine tablets are a nucleoside analogue reverse transcriptase
inhibitor indicated in combination with other antiretroviral agent for the treatment of HIV-1
infection.
Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Valsartan Tablets USP - AB Rated to Diovan® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Valsartan Tablets USP, 40mg, 80mg, 160mg, and 320mg. Lupins new product is the AB-rated generic equivalent of Novartis Corporations, Diovan® Tablets.
Lupins Valsartan Tablets USP is an angiotensin II receptor blocker (ARB) indicated for:
- Treatment of hypertension, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions (1.1)
- Treatment of heart failure (NYHA class II-IV); valsartan tablets USP significantly reduced hospitalization for heart failure (1.2)
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Telmisartan & Hydrochlorothiazide Tablets USP - AB rated to Micardis® HCT
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Telmisartan and
hydrochlorothiazide tablets USP. Lupins Telmisartan hydrochlorothiazide tablets USP are the
AB-rated generic equivalent of Boehringer Ingelheims Micardis® HCT.
Telmisartan and hydrochlorothiazide tablets USP are indicated for the treatment of hypertension.
This fixed dose combination is not indicated for initial therapy.
The Company has already commenced shipment of the product in the U.S. Please contact your
Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Nikki™ Tablets - AB rated to Yaz®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Nikki™ Tablets (drospirenone and ethinyl estradiol tablets USP), 3mg/0.02mg. Lupin's NikkiTM is the AB-rated generic equivalent of Bayer's Yaz® and is indicated for use by women to prevent pregnancy.
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Ciprofloxacin for Oral Suspension - AB rated to Cipro® OS
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Ciprofloxacin for oral suspension, 250mg/5mL and 500mg/5mL.
Lupin's Ciprofloxacin for oral suspension is the AB-rated generic equivalent of Bayer's Cipro® oral suspension and is indicated for the treatment of infections caused by susceptible isolates of
the designated microorganisms in various conditions and patient populations.
Lupin is the first applicant to file an ANDA for Cipro® oral suspension 250mg/5mL & 500mg/
5mL and as such is entitled to 180 days of marketing exclusivity. The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Zolpidem Tartrate ER Tablets USP -- AB rated to Ambien® CR
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Zolpidem Tartrate Extended-Release Tablets USP, 6.25mg and 12.5mg.
Lupin's Zolpidem Tartrate ER Tabletsare the AB-rated generic equivalent of Sanofi-Aventis' Ambien® CR and are indicated for the treatment of insomnia characterized by difficulties with sleep onset and/or sleep maintenance.
Lupin has commenced shipment of this product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Eszopiclone Tablets - AB rated to Lunesta®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Eszopiclone tablets, 1mg, 2mg & 3mg.
Lupin's Eszopiclone tablets are the AB-rated generic equivalent of Sunovion's Lunesta® tablets. Eszopiclone tablets are indicated for the treatment of insomnia and have been shown to decrease sleep latency and improve sleep maintenance.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Niacin ER Tablets USP - AB rated to Niaspan®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Niacin ER tablets USP, 500mg, 750mg & 1000mg.
Lupin's Niacin ER tablets USP are the AB-rated generic equivalent of Abbvie's Niaspan®.
Niacin ER tablets USP contain extended-release Niacin (nicotinic acid), and is indicated:
- To reduce elevated TC, LDL-C, Apo B and TG, and to increase HDL-C in patients with primary hyperlipidemia and mixed dyslipidemia.
- In combination with simvastatin or lovastatin: to treat primary hyperlipidemia and mixed dyslipidemia when treatment with niacin extended-release tablets USP, simvastatin, or lovastatin monotherapy is considered inadequate.
- To reduce the risk of recurrent nonfatal myocardial infarction in patients with a history of myocardial infarction and hyperlipidemia.
- To reduce TG in adult patients with severe hypertriglyceridemia.
- In combination with a bile acid binding resin:
- Slows progression or promotes regression of atherosclerotic disease in patients with a history of coronary artery disease (CAD) and hyperlipidemia.
- As an adjunct to diet to reduce elevated TC and LDL-C in adult patients with primary hyperlipidemia
Lupin has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Rifabutin Capsules USP - AB rated to Mycobutin® Capsules USP
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Rifabutin Capsules USP, 150mg. Rifabutin Capsules are the AB-rated generic equivalent of Pfizer's Mycobutin® Capsules.
Rifabutin Capsules are indicated for the prevention of disseminated Mycobacterium avium complex (MAC) disease in patients with advanced HIV infection.
Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Vyfemla™ - AB rated to Ovcon® 35
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Vyfemla™ (Norethindrone and Ethinyl Estradiol Tablets USP 0.4 mg/0.035 mg).
Lupin's Vyfemla™ is the AB-rated generic equivalent of Warner Chilcott's Ovcon® 35. Vyfemla™ is an oral contraceptive that is indicated for the prevention of pregnancy in women who elect to use this product as a method of contraception.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Telmisartan & Amlodipine Tablets - AB rated to Twynsta®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Telmisartan and Amlodipine Tablets. Lupin's Telmisartan and Amlodipine Tablets are the AB-rated generic equivalent of BoehringerIngelheim's Twynsta®.
Telmisartan and Amlodipine Tablet is an angiotensin II receptor blocker (ARB) and a dihydropyridine calcium channel blocker (DHP-CCB) combination product indicated for the following:
- The treatment of hypertension alone or with other antihypertensive agents to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions.
- Initial therapy inpatients likely to need multiple antihypertensive agents to achieve their blood pressure goals
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Abacavir Sulfate, Lamivudine, and Zidovudine Tablets - AB rated to Trizivir®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Abacavir Sulfate, Lamivudine, and Zidovudine Tablets, 300mg (base) / 150mg / 300mg.
Lupin's Abacavir Sulfate, Lamivudine ZidovudineTablets 300mg (base)/150mg/ 300mg are the AB-rated generic equivalent of ViiV Healthcare's (ViiV) Trizivir® Tablets, 300mg (base) / 150mg / 300mg and are indicated in combination with other antiretrovirals or alone for the treatment of HIV-1 infection.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Duloxetine DR Capsules- AB rated to Cymbalta®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Duloxetine Delayed-Release Capsules. Lupin's Duloxetine is AB-rated generic equivalent of Eli Lilly's Cymbalta®.
Duloxetine Delayed-Release Capsules USP are indicated for:
- the treatment of major depressive disorders (MDD)
- the treatment of generalized anxiety disorder (GAD)
- the management of neuropathic pain (DPNP) associated with diabetic peripheral neuropathy
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Rifampin Capsules 150mg & 300mg - AB rated to Rifadin®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Rifampin Capsules. Lupin's Rifampin Capsules are the AB-rated generic equivalent of Sanofi Aventis' Rifadin®. Rifampin is indicated in the treatment of all forms of tuberculosis.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Fenofibric Acid Delayed-Release Capsules 45mg & 135mg - AB rated to Trilipix®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Fenofibric Acid delayed-release capsules. Lupin's Fenofibric Acid DR capsules are the AB-rated generic equivalent of Abbott LaboratoriesTrilipix®.
Fenofibric Acid delayed-release capsules are indicated as an adjunct to diet in combination with a statin to reduce TG and increase HDL-C in patients with mixed dyslipidemia and CHD or a CHD risk equivalent who are on optimal statin therapy to achieve their LDL-C goal.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Rabeprazole Na DR Tablets - AB rated to Aciphex®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Rabeprazole Na DR Tablets. Lupin's Rabeprazole Na DR Tablets is the AB-rated generic equivalent Aciphex®. Rabeprazole Na DR Tabs is a proton pump inhibitor indicated for the treatment of heartburn, stomach ulcers, gastroesophageal reflux disease (GERD), esophagus damage and conditions that cause excessive stomach acid, such as Zollinger-Ellisonsyndrome.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Gatifloxacin Ophthalmic Solution 0.5% - AT rated to Zymaxid®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Gatifloxacin Ophthalmic Solution, 0.5%. Lupin's Gatifloxacin O.S. is the AT-rated generic equivalent of Zymaxid®. Gatifloxacin O.S. is a topical fluoroquinolone anti-infective indicated for the treatment of bacterial conjunctivitis.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Pirmella™ 1/35--AB rated to Ortho-Novum® 1/35
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Pirmella™ 1/35 (Norethindrone and Ethinyl Estradiol Tablets USP 1mg/0.035mg). Lupin's Pirmella™ 1/35 is the AB-rated generic equivalent of Ortho-Novum® 1/35.
Pirmella™ 1/35 is indicated for the prevention of pregnancy in women who elect to use this product as a method of contraception.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
 |
Lupin Launches Pirmella™ 7/7/7--AB rated to Ortho-Novum® 7/7/7
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Pirmella™ 7/7/7 (Norethindrone and Ethinyl Estradiol Tablets USP - 0.5 mg/0.035 mg; 0.75 mg/0.035 mg and 1 mg/0.035 mg). Lupin's Pirmella™ 7/7/7 is the AB-rated generic equivalent of Ortho-Novum® 7/7/7.
Pirmella™ 7/7/7 is indicated for the prevention of pregnancy in women who elect to use this product as a method of contraception.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
 |
Lupin Launches Doxycycline for Oral Suspension USP-AB rated to Vibramycin®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Doxycycline for Oral Suspension USP. Lupin's Doxycycline for Oral Suspension USP is the AB-rated generic equivalent Vibramycin®. Doxycycline is a broad-spectrum antibiotic with multiple indications for treatment.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Jencycla™ (Norethindrone Tablets USP 0.35 mg-AB rated to Ortho-Micronor®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Jencycla™ (Norethindrone Tablets USP 0.35 mg). Lupin's Jencycla™ is the AB-rated generic equivalent Ortho-Micronor®. Jencycla™ is an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
Lupin Launches Enskyce™ (Desogestrel and Ethinyl Estradiol Tablets USP 0.15 mg/0.03 mg) - AB rated to Ortho-Cept®.
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Enskyce™ (Desogestrel and Ethinyl Estradiol Tablets USP 0.15 mg/0.03 mg). Enskyce™ is an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
||||||||||||||||
Lupin Launches Daysee™ (Levonorgestrel and Ethinyl Estradiol Tablets USP 0.15 mg/0.03 mg and Ethinyl Estradiol Tablets USP 0.01 mg) - AB rated to Seasonique®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Daysee™ (Levonorgestrel and Ethinyl Estradiol Tablets USP 0.15 mg/0.03 mg and Ethinyl Estradiol Tablets USP 0.01 mg). Daysee™ is an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
||||||||||||||||||
Lupin Launches Irbesartan and Hydrochlorothiazide Tablets, AB rated to Avalide®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Irbesartan and Hydrochlorothiazide Tablets, 150mg/12.5mg and 300mg/12.5mg. Irbesartan and Hydrochlorothiazide Tablets are the AB-rated generic equivalent of UCB Pharma's Avalide® and is an angiotensin II receptor antagonist with a diuretic indicated for the treatment of hypertension.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
 |
| SDS | |
| Package Insert |
Lupin Launches Valsartan & Hydrochlorothiazide Tablets USP, AB Rated to Diovan HCT® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Valsartan and Hydrochlorothiazide Tablets USP, 80mg/12.5mg, 160mg/12.5mg, 160mg/25mg, 320mg/12.5mg and 320mg/25mg. Lupin's new product is the AB-rated generic equivalent of Novartis Corporations, Diovan HCT® Tablets.
Lupin's Valsartan and Hydrochlorothiazide Tablets USP are indicated for the treatment of hypertension, to lower blood pressure in patients not adequately controlled with monotherapy or as initial therapy in patients who are likely to need multiple drugs to achieve their blood pressure goals.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
||||||||||||||||||
Lupin Launches Levonorgestrel and Ethinyl Estradiol Tablets USP 0.1 mg/0.02 mg (EQ of Lutera®)
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Levonorgestrel and Ethinyl Estradiol Tablets USP 0.1 mg/0.02 mg. Lupin's Levonorgestrel and Ethinyl Estradiol Tablets USP 0.1 mg/0.02 mg are the AB-rated generic equivalent of Watson Pharmaceuticals, Lutera®. This product is an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
||||||||||||||||||
Lupin Launches Fenofibrate Capsules, 43mg & 130mg - AB rated to Antara®
Lupin Pharmaceuticals, Inc. is proud to announce that it has commenced commercial shipments of Fenofibrate Capsules, 43mg and 130mg. Lupin's Fenofibrate Capsule is the authorized generic of Lupin's Antara®. Fenofibrate Capsules are indicated as adjunctive therapy to diet to reduce elevated low-density lipoprotein cholesterol (LDL-C), total cholesterol (Total-C), triglycerides (TG), and apolipoprotein B (Apo B), and to increase high density lipoprotein cholesterol (HDL-C) in adult patients with primary hyperlipidemia or mixed dyslipidemia.
Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
| SDS | |
| Package Insert | |
Lupin Launches Desloratadine Tablets 5mg, AB rated to Clarinex®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Desloratadine Tablets, 5mg. Lupin's Desloratadine tablets are the AB-rated generic equivalent of Schering Corporations, Clarinex®. Desloratadine tablets are indicated for the relief of the nasal and non-nasal symptoms of seasonal allergic rhinitis in patients 2 years of age and older.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
||||||||||||||||||
Lupin Launches Levetiracetam Oral Solution USP, AA rated to Keppra® OS
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Levetiracetam Oral Solution USP 100mg/mL x 473mL. Levetiracetam Oral Solution is the AA-rated generic equivalent of UCB's Keppra® OS and is an antiepileptic indicated for the treatment of seizures.
Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
||||||||||||||||||
Lupin Launches Drospirenone & Ethinyl Estradiol Tablets USP 3mg/0.03mg, AB rated to Yasmin®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Drospirenone & Ethinyl Estradiol Tablets USP 3mg/0.03mg. Lupin's Drospirenone & Ethinyl Estradiol Tablets USP 3mg/0.03mg are the AB-rated generic equivalent of Bayer Schering Pharma's Yasmin®. Drospirenone & Ethinyl Estradiol Tablets USP are an oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
 |
||||||||||
Lupin Launches Fenofibrate Tablets, AB rated to Tricor®
Lupin Pharmaceuticals Inc. is proud to announce its recent product launch, Fenofibrate Tablets, 48mg and 145mg. Lupin's Fenofibrate is a peroxisome proliferator receptor alpha (PPARα) activator indicated as an adjunct to diet to reduce elevated LDL-C, Total-C, TG and Apo B, and to increase HDL-C in adult patients with primary hypercholesterolemia or mixed dyslipidemia and for treatment of adult patients with severe hypertriglyceridemia.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
||||||||||||||||||
Lupin receives FDA approval for Generic Nordette® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Kurvelo™ (Levonorgestrel and Ethinyl Estradiol Tablets, USP 0.15 mg/0.03 mg). Lupin's Kurvelo™ Tablets are the AB-rated generic equivalent of Wyeth, LLC's Nordette® Tablets. Lupin's Kurvelo™ Tablets are a combined oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception. Kurvelo™ is a registered trademark of Lupin Pharmaceuticals, Inc.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
||||||||||||||||||||
Lupin receives FDA approval for Generic Avapro® Tablets
Lupin Pharmaceuticals Inc. is proud to announce its recent product launch, Irbesartan Tablets, 75mg, 150mg, and 300mg. Lupin's Irbesartan Tablets is the AB-rated generic equivalent of Sanofi Corporations Avapro® Tablets and is an angiotensin II receptor antagonist used to treat hypertension.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
||||||||||||||||||||
Lupin receives FDA approval for Generic Seasonale® Tablets
Lupin Pharmaceuticals Inc. is proud to announce its recent product launch, Levonorgestrel and Ethinyl Estradiol Tablets, USP 0.15 mg/0.03 mg. Lupin's Levonorgestrel and Ethinyl Estradiol Tablets, USP 0.15 mg/0.03 mg are the AB-rated generic equivalent of Teva's Seasonale® Tablets. Lupin's Levonorgestrel and Ethinyl Estradiol tablets are a combined oral contraceptive indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
||||||||||||||||||||
Lupin receives FDA approval for Generic Lexapro® Tablets
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Escitalopram Oxalate Tablets, 5 mg, 10 mg, and 20 mg. Lupin's Escitalopram Oxalate Tablets is the AB-rated generic equivalent of Forest's Lexapro® Tablets and is indicated for acute and maintenance treatment of major depressive disorder in adults and adolescents aged 12-17 years. It is also indicated for the acute treatment of general anxiety disorder in adults.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
||||||||||||||||||||
Lupin Launches Lamivudine and Zidovudine Tablets, AB rated to Combivir®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Lamivudine and Zidovudine Tablets, 150mg/300mg. Lamivudine and Zidovudine tablet, a combination of two nucleoside analogue reverse transcriptase inhibitors, is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
||||||||||||||||||||
Lupin Resumes Sales of Metformin ER Tablets, AB rated to Fortamet®
The U.S. Court of Appeals for the Federal Circuit has granted Lupin's request to stay the preliminary injunction that had earlier barred sales of its generic of Shionogi's Fortamet® tablets.
Lupin had earlier received final approval for its Metformin tablets from the U.S. FDA and had launched the product in September, 2011. In December, 2011 Shionogi's request for a preliminary injunction had been granted and had blocked Lupin from further sales of the product.
Fortamet® is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
The Company has resumed shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
 |
| SDS | |
| Package Insert | |
| Medication Guide | |
Lupin Launches Quetiapine Fumarate Tablets, AB rated to Seroquel®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Quetiapine Fumarate Tablets, 25mg, 50mg, 100mg, 200mg, 300mg and 400mg. Quetiapine fumarate is an atypical antipsychotic drug indicated for the treatment of schizophrenia and bipolar disease.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
||||||||||||||||||||||
Lupin Launches Ziprasidone Hydrochloride Capsules, AB rated to Geodon®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Ziprasidone Hydrochloride Capsules, 20mg, 40mg, 60mg and 80mg. Ziprasidone HCl is an atypical antipsychotic drug indicated for the treatment of schizophrenia.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
||||||||||||||||||||||
WYMZYA™ Fe (previously norethindrone and ethinyl estradiol tablets, chewable and ferrous fumarate tablets 0.4mg/35mcg) now available, AB Rated to Femcon® Fe
Lupin Pharmaceuticals, Inc. is proud to announce that it has commenced commercial shipments of WYMZYA™ Fe. Previously, this product was shipped under the established name of norethindrone and ethinyl estradiol tablets, chewable and ferrous fumarate tablets 0.4mg/35mcg. WYMZYA™ Fe is the authorized generic of Femcon® Fe and is a combination oral contraceptive.
Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|
||||||||||||||||||||||
Lupin Launches Levonorgestrel and Ethinyl Estradiol Tablets USP (0.1 mg/0.02 mg) and Ethinyl Estradiol Tablets USP (0.01 mg), AB Rated to LoSeasonique®
Lupin Pharmaceuticals, Inc. is proud to announce its recent product launch, Levonorgestrel and Ethinyl Estradiol Tablets USP (0.1 mg/0.02 mg) and Ethinyl Estradiol Tablets USP (0.01 mg), AB rated to LoSeasonique®. Levonorgestrel and Ethinyl Estradiol Tablets USP (0.1 mg/0.02 mg) and Ethinyl Estradiol Tablets USP (0.01 mg) is a combination oral contraceptive.
The Company has already commenced shipment of the product in the U.S. Please contact your Lupin Sales Representative or call 1-866-587-4617 for ordering information.
|
|
|




















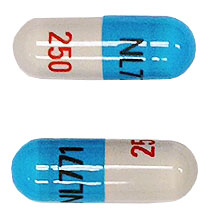





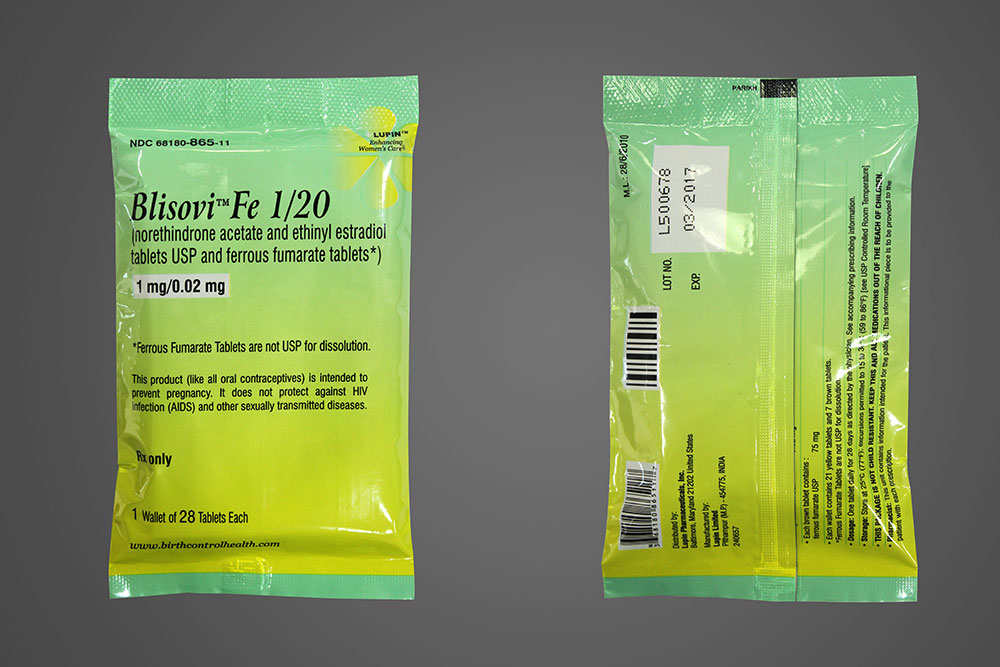

.jpg)
.jpg)