PRODUCTS
| Product Name | Strength | Image (not to scale) | Pack Size | NDC # | RLD / Brand Name | TE Rating | Therapeutic Category |
|---|---|---|---|---|---|---|---|
| DOXOrubicin Hydrochloride Liposome Injection | 20mg/10mL (2mg/mL) |
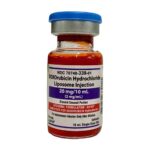 |
(1) 10mL single-dose vial |
70748-339-01 |
Doxil® |
AB |
Chemotherapeutic Agents |
| GaviLyte™-N | 420-111.20-1.48-5.72 gm |
 |
4 L |
43386-050-19 |
NuLYTELY® |
AA |
Hyperosmotic |
| Tiotropium Bromide Inhalation Powder Capsules | 18mcg / capsules |
 |
30 |
68180-964-12 |
Spiriva® HandiHaler® |
AB |
Anticholinergics |
| Rufinamide Oral Suspension | 40 mg/mL |
 |
460ml |
68180-797-01 |
Banzel® |
AB |
Antiepileptic |
| Meloxicam Capsules | 5mg |
 |
30 |
68180-188-06 |
Vivlodex® |
AB |
analgesic |
| Arformoterol Tartrate Inhalation Solution | 15mcg/2mL |
 |
30 UD Vials |
70748-0257-30 |
Brovana® |
NA |
Long-acting beta agonists (LABAs) |
| Penicillamine Tablets USP | 250 mg |
 |
100 |
70748-0153-01 |
Depen® |
AB |
Antirheumatic |
| Posaconazole Delayed-Release Tablets | 100mg |
 |
60 |
70748-0258-07 |
Noxafil® |
AB |
Antifungal |
| Lapatinib Tablets | 250 mg |
 |
150 |
68180-0801-36 |
Tykerb® |
AB |
Antineoplastic Agent |
| Fosaprepitant Dimeglumine for Injection | 150mg |
 |
5mL single-dose vial |
68180-690-01 |
Emend® |
AP |
Antiemetic |
| Divalproex Sodium Extended-Release Tablets USP | 250mg |
 |
100 |
68180-260-01 |
Depakote® ER |
AB |
Anti-Convulsant |
| Doxycycline Hyclate Tablets | 75mg |
 |
60 |
68180-653-07 |
Acticlate |
AB |
Tetracycline Antibiotics |
| Leflunomide Tablets USP | 10 mg |
 |
30 |
70748-129-06 |
Arava® |
AB |
Immune Suppressant |
| Albuterol Sulfate Inhalation Aerosol | 90 mcg per actuation |
 |
8.5g |
68180-963-01 |
PROAIR® HFA |
AB2 |
Bronchodilator |
| Zileuton Extended-Release Tablets | 600mg |
 |
120 |
68180-169-16 |
Zyflo CR® |
AB |
Anti-inflammatory |
| Drospirenone, Ethinyl Estradiol & Levomefolate Calcium & Levomefolate Calcium Tablets | 3mg/0.02mg/0.451mg and 0.451mg |
 |
3 x 28 |
68180-0894-73 |
Beyaz® |
AB |
Oral Contraceptives |
| Rifabutin Capsules USP | 150mg |
 |
100 |
68180-0285-01 |
Mycobutin® |
AB |
Antitubercular |
| Voriconazole Oral Suspension | 40 mg/mL |
 |
49 g (75 mL when reconstituted) |
43386-0038-60 |
Vfend® |
AB |
Antifungal |
| Vyfemla™ | 0.4mg/0.035mg |
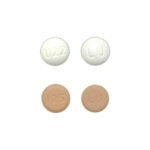 |
28 x 3 |
68180-0875-73 |
Ovcon® 35 |
AB |
Oral Contraceptive |
| Wymzya™ | 0.4mg/35mg |
 |
28 x 3 |
68180-0873-73 |
Femcon® Fe |
AB |
Oral Contraceptive |
| Ziprasidone HCl Capsules | 20mg |
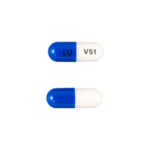 |
60 |
68180-331-07 |
Geodon® |
AB |
Antipsychotic |
| Zolpidem Tartrate ER Tablets USP | 6.25mg |
 |
100 |
68180-779-04 |
Ambien® CR |
AB |
Sedative/Hypnotic |
| Tri-Lo-Marzia™ | 0.18mg/0.025mg |
 |
28 x 3 |
68180-0837-73 |
Ortho Tri-Cyclen® Lo |
AB |
Oral Contraceptive |
| Tobramycin Inhalation Solution USP 300mg/ 5mL | 300mg/ 5mL |
 |
56 Single-Use Ampules |
68180-0962-56 |
Tobi® |
AN |
Antibacterial |
*All registered trademarks are the property of their respective owners. These products are intended for U.S. residents only.
