PRODUCTS
| Product Name | Strength | Image (not to scale) | Pack Size | NDC # | RLD / Brand Name | TE Rating | Therapeutic Category |
|---|---|---|---|---|---|---|---|
| Cephalexin Capsules USP | 250 mg |
 |
100 |
68180-121-01 |
Keflex® |
AB |
Cephalosporin Antibiotic |
| Celecoxib Capsules | 50 mg |
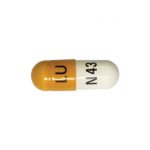 |
60 |
68180-395-07 |
Celebrex® |
AB |
Anti-arthritic |
| Cefprozil Tablets USP | 250 mg |
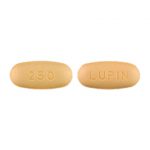 |
50 |
68180-403-01 |
Cefzil® |
AB |
Cephalosporin Antibiotic |
| Cefprozil for Oral Suspension USP | 125 mg/5 mL |
 |
50 mL |
68180-401-01 |
Cefzil® |
AB |
Cephalosporin Antibiotic |
| Cefixime for Oral Suspension USP | 100 mg/5 mL |
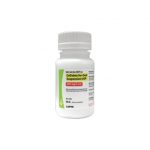 |
50 mL |
68180-405-01 |
Suprax® |
AB |
Cephalosporin Antibiotic |
| Atovaquone Oral Suspension USP | 750 mg/5 mL |
 |
210 mL |
70748-299-01 |
Mepron® |
AB |
Antiprotozoal |
| Blisovi™ Fe 1.5/30 Tablets USP | 1.5 mg/0.03 mg |
 |
28 x 3 |
68180-866-73 |
Loestrin® Fe 1.5/30 |
AB |
Oral Contraceptives |
| Blisovi™ Fe 1/20 Tablets USP | 1 mg/0.02 mg |
 |
28 x 3 |
68180-865-73 |
Loestrin® Fe 1/20 |
AB |
Oral Contraceptives |
| Blisovi™ 24 Fe Tablets USP | 1 mg/0.02 mg |
 |
28 x 3 |
68180-864-73 |
Loestrin® 24 Fe |
AB |
Oral Contraceptives |
| Cefixime Capsules | 400 mg |
 |
1 x 10 |
68180-423-11 |
Suprax® |
AB |
Antibacterial |
| Cefdinir for Oral Suspension | 125 mg/5 mL |
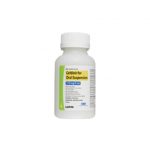 |
60 mL |
68180-722-04 |
Omnicef® |
AB |
Cephalosporin |
| Cefdinir Capsules | 300 mg |
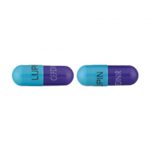 |
60 |
68180-711-60 |
Omnicef® |
AB |
Cephalosporin |
*All registered trademarks are the property of their respective owners. These products are intended for U.S. residents only.
